Miokarditis dan perikarditis telah dilaporkan sebagai salah satu efek samping pasca vaksinasi COVID-19 jenis mRNA, baik Pfizer-BioNTech maupun Moderna. Terdapat laporan adanya peningkatan kasus miokarditis dan perikarditis pada usia muda setelah vaksinasi COVID-19 di Amerika Serikat sejak April 2021. Sedangkan European Medicines Agency’s (EMA) safety committee, yaitu The Pharmacovigilance Risk Assessment Committee (PRAC), menyebutkan bahwa miokarditis dan perikarditis adalah kejadian yang sangat jarang setelah vaksin mRNA COVID-19.[1,2]
Vaksin COVID-19 mRNA
Vaksin COVID-19 mRNA yang tersedia dan digunakan di Indonesia yaitu vaksin mRNA-1273 (vaksin Moderna atau SpikeVax) dan vaksin BNT162b2-mRNA (vaksin Pfizer atau Corminaty). Kedua vaksin ini bekerja dengan cara memberikan respon imun terhadap antigen spike (S) pada severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2).[3-6]
Vaksin Moderna diberikan pada usia >8 tahun (2 dosis pemberian dan interval 28 hari), sedangkan vaksin Pfizer dapat diberikan pada usia >12 tahun (2 dosis pemberian dan interval 21 hari). Uji interim pada fase 3 menunjukkan efikasi vaksin Moderna pada usia 18 hingga <65 tahun sebesar 95,6%, dan efikasi vaksin Pfizer pada usia >16 tahun sebesar 95,5%.[3-6]
Berdasarkan keputusan Kementerian Kesehatan Republik Indonesia (Kemenkes RI), vaksin Moderna saat ini juga digunakan sebagai dosis ketiga atau booster untuk tenaga kesehatan di Indonesia.[3-6]
Studi Kejadian Miokarditis dan Perikarditis Setelah Vaksinasi COVID-19 mRNA
Miokarditis merupakan inflamasi pada otot jantung, sedangkan perikarditis adalah inflamasi pada lapisan luar jantung. Kejadian miokarditis dan perikarditis sebenarnya telah dilaporkan sebagai efek samping yang jarang setelah vaksinasi terdahulu, misalnya pada vaksin influenza, vaksin hepatitis B, dan vaksin varicella.[7,8]
Beberapa studi dan laporan insidensi terkait miokarditis dan perikarditis setelah vaksin COVID-19 mRNA adalah:
- Studi Diaz et al: dari sekitar >2 juta individu yang mendapatkan vaksin Pfizer atau moderna, dilaporkan 20 orang mengalami miokarditis dan 37 orang mengalami perikarditis.[9]
- Studi Montgomery et al: dari total 2,8 juta dosis vaksinasi pada personel militer di departemen pertahanan Amerika Serikat, dilaporkan 23 orang mengalami miokarditis.[10]
- Data Kementerian Kesehatan di Kanada hingga 9 Juli 2021: dari total >41 juta dosis vaksin COVID-19 yang telah disuntikkan, dilaporkan 163 pasien mengalami miokarditis dan/atau perikarditis. Di mana sebanyak 111 pasien pasca vaksin Pfizer dan 40 pasien pasca vaksin Moderna.[13]
- Data EMA sampai 31 Mei 2021: dari sekitar 177 juta dosis vaksin Pfizer dan 20 juta dosis vaksin Moderna yang telah diberikan di European Economic Area (EEA), dilaporkan miokarditis sebanyak 145 kasus pasca vaksin Pfizer dan 19 kasus pasca vaksin Moderna.[1]
- Data Centers For Disease Control and Prevention (CDC): memperkirakan angka kejadian miokarditis atau perikarditis sebesar 12,6 kasus/juta dosis, setelah dosis kedua vaksin mRNA, di antara individu usia 12−39 tahun, pada 21 hari setelah penyuntikan.[14]
Insidensi Miokarditis
Secara global, insidensi miokarditis bukan karena vaksinasi dilaporkan sekitar 10−20 kasus/100.000 orang/tahun. Oleh karena itu, masih dibutuhkan studi tambahan untuk mengonfirmasi apakah kejadian miokarditis setelah vaksinasi COVID-19 memang lebih tinggi dibandingkan pada populasi umum. Namun, dengan adanya kejadian ini tetap perlu mendapat perhatian khusus bagi tenaga kesehatan maupun pasien yang mendapatkan vaksin mRNA.[1,16]
Risiko Miokarditis dan Perikarditis Akibat Vaksin COVID-19 mRNA
Studi Diaz et al. menyatakan bahwa miokarditis pasca vaksin COVID-19 mRNA terjadi dalam beberapa hari pasca vaksin, yaitu sekitar 3 hari setelah penyuntikan. Sedangkan, perikarditis pasca vaksin COVID-19 mRNA terjadi dalam waktu yang lebih lama daripada miokarditis, yaitu 20 hari setelah penyuntikan.[9]
Laki-laki Usia Remaja dan Dewasa Muda
Sebagian besar kasus melaporkan miokarditis dan perikarditis pasca vaksinasi COVID-19 ini terjadi pada remaja dan dewasa muda <30 tahun, serta lebih sering pada laki-laki daripada wanita. Miokarditis berkembang secara cepat pada pasien usia muda, sedangkan perikarditis mengenai pasien usia lebih tua. Selain itu, menurut Diaz et al, miokarditis terjadi 75% pada laki-laki dengan umur 36 tahun.[9,17]
Studi terbaru yang dipublikasikan pada tanggal 15 Desember 2021, memperlihatkan risiko miokarditis dari vaksin COVID-19. Hasil studi menunjukkan pada populasi usia <40 tahun, risiko miokarditis setelah suntikan kedua vaksin moderna lebih tinggi daripada risiko miokarditis akibat COVID-19. Bahkan, perbedaan tersebut tampaknya lebih tinggi lagi karena inklusi penderita COVID-19 berdasarkan hasil tes PCR, sehingga pasien tanpa gejala atau gejala ringan COVID-19 tidak dimasukan ke dalam studi.[33]
Efek samping vaksin Pfizer pada populasi yang lebih luas dilaporkan berdasarkan database negara Israel. Peningkatan risiko miokarditis terutama terjadi pada remaja dan dewasa muda usia 16−39 tahun. Namun, peningkatan risiko tersebut tidak sebanding dengan risiko yang sama akibat terinfeksi COVID-19.[34]
Dosis Kedua Vaksin COVID-19 mRNA
Risiko miokarditis atau perikarditis tampaknya lebih tinggi setelah dosis kedua vaksin COVID-19 mRNA. Namun, terdapat beberapa kasus yang dilaporkan setelah dosis pertama. Sebesar 76% kasus yang dilaporkan ke vaccine adverse event reporting system (VAERS) di Amerika Serikat terjadi setelah dosis kedua. Menurut CDC, miokarditis lebih sering terjadi pasca dosis kedua, sedangkan perikarditis terjadi setelah dosis pertama maupun dosis kedua.[8,10,18]
Mekanisme Potensial Miokarditis dan Perikarditis pasca Vaksinasi COVID-19 mRNA
Hingga saat, mekanisme pasti inflamasi jantung setelah vaksin COVID-19 mRNA masih terus diteliti. Vaksin mRNA SARS-CoV-2 mengandung nucleoside-modified mRNA yang mengkode glikoprotein dari spike virus. Vaksin mRNA di dalam sel inang membangun protein spike yang merangsang respons imun adaptif untuk mengidentifikasi dan menghancurkan virus. Antibodi spike protein IgG akan mencegah perlekatan virus SARS-CoV-2 ke sel inang, melalui ikatan protein spike pada reseptor angiotensin-converting enzyme 2, sehingga menetralkan virus.[19]
Mekanisme Autoantibodi Reaktif Jantung
Berdasarkan satu laporan kasus, pasien dengan miokarditis mengalami peningkatan kadar antagonis reseptor interleukin-1 (IL-1), IL-5, IL-16, tetapi bukan sitokin proinflamasi seperti IL-6, faktor nekrosis tumor, IL-1B, IL-2, atau interferon-γ (IF-γ). Pada pasien tersebut juga memiliki tingkat antibodi yang lebih tinggi terhadap self-antigen, termasuk aquaporin 4, endothelial cell antigen, dan protein proteolipid. Pasien dengan miokarditis memiliki autoantibodi reaktif jantung yang lebih tinggi. Autoantibodi reaktif jantung dapat melawan antigen multipel, beberapa di antaranya mungkin memiliki efek fungsional pada miosit jantung. Pembentukan autoantibodi ini dapat menjadi salah satu mekanisme miokarditis dapat berkembang pada individu yang rentan setelah vaksinasi.[20-22]
Mekanisme Mimikri Molekuler
Mekanisme potensial miokarditis lain yang penting adalah mimikri molekuler antara protein spike SARS-CoV-2 dan self-antigen. Antibodi terhadap glikoprotein spike SARS-CoV-2 telah terbukti secara eksperimental bereaksi silang dengan sekuens protein peptida manusia yang serupa secara struktural, termasuk miosin.[23,24]
Meskipun vaksinasi COVID-19 tampaknya tidak memprovokasi efek samping yang dimediasi imun de novo, tetapi ada kemungkinan dapat memicu jalur disregulasi yang sudah ada sebelumnya pada individu tertentu. Kondisi ini akan menghasilkan ekspansi sel B poliklonal, membentuk kompleks imun, dan menyebabkan peradangan.[23]
Mekanisme Testosteron dan Estrogen terhadap Respon Imun
Penyebab dominasi laki-laki dalam kasus miokarditis atau perikarditis belum diketahui pasti. Penjelasan yang dapat dikaitkan adalah perbedaan hormon seks, di mana hormon testosteron diduga berperan dalam mekanisme penghambatan sel anti-inflamasi.[25-27]
Sedangkan hormon estrogen memiliki efek menghambat sel T proinflamasi, sehingga menurunkan respon imun yang diperantarai sel. Faktor lain yang mungkin berkontribusi adalah kurangnya diagnosis pada wanita.[28,19]
Presentasi Klinis Miokarditis dan Perikarditis Pasca Vaksinasi COVID-19 mRNA
Gejala dan tanda klinis miokarditis dan perikarditis pasca vaksinasi Moderna rata-rata timbul pada hari ke-3, dan paling sering pada hari ke-2 setelah penyuntikan. Sebagian besar dilaporkan terjadi pada laki-laki, dengan usia 16−18 tahun.[16]
Secara umum, gejala yang terjadi berupa nyeri dada, palpitasi, sesak napas, demam, lemah, mual, muntah, dan nyeri perut. Tanda klinis umum termasuk takipnea, takikardia, murmur, gallop, bradikardia, hipotensi, hepatomegali, edema, dan tanda-tanda curah jantung rendah.[16]
Berdasarkan studi Das et al., gejala miokarditis dan perikarditis yang paling sering terjadi adalah nyeri dada. Gejala muncul pada hari ke-1 hingga ke-7, terbanyak pada hari ke-3 setelah penyuntikan. Beberapa pasien didahului dengan demam dan mialgia 1 hari setelah penyuntikan.[29]
Diagnosis Miokarditis dan Perikarditis Pasca Vaksinasi COVID-19 mRNA
CDC memantau 484 kasus dugaan miokarditis atau perikarditis pada pasien usia 29 tahun, dan melaporkan bahwa 86% pasien datang dengan keluhan nyeri dada. Hasil pemeriksaan penunjang menunjukkan bahwa 61% pasien memiliki perubahan gelombang ST atau T pada elektrokardiogram (EKG), 64% pasien mengalami peningkatan enzim jantung troponin pada hari ke-3 setelah vaksin, dan 17% memberikan gambaran pencitraan jantung abnormal.[14]
Pemeriksaan Khusus Jantung
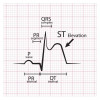
Pada sekitar 85% individu memiliki gambaran EKG abnormal dengan adanya ST-elevasi di inferior leads. Terdapat gelombang QRS melebar yang menandakan sistem konduksi bermasalah, blok atrioventrikular, sinus takikardia, aritmia ventrikel, atau bradikardia simptomatik.[11]
Laporan kasus oleh Ammirati et al., terdapat laki-laki berusia 56 tahun yang mengalami nyeri dada dengan onset akut 3 hari pasca dosis kedua vaksin Pfizer. Hasil EKG menunjukkan hasil irama sinus dengan elevasi ST yang minimal pada precordial leads dan gelombang T yang tinggi. Hasil yang sama juga ditunjukkan oleh Das et al., gambaran EKG abnormal paling sering ditemukan adalah elevasi gelombang ST sebanyak 38%.[29,30]
Pada pemeriksaan ekokardiografi menunjukkan adanya disfungsi yang ringan pada 13 dari 29 pasien. Pada pasien dengan usia >30 tahun, sebanyak 66% mengalami penurunan fungsi ventrikel kiri. Sedangkan pada usia <30 tahun, fungsi ventrikel kiri menurun pada 34% pasien.[29]
Pemeriksaan Laboratorium
Pemeriksaan laboratorium yang digunakan meliputi pemeriksaan untuk mendeteksi virus penyebab miokarditis, konsentrasi serum penanda inflamasi (protein C-reaktif, laju sedimentasi eritrosit), biomarker jantung (troponin, peptida natriuretik tipe-B), dan uji serologi antibodi SARS-CoV-2.[16]
Level peptida natriuretik tipe-B hanya meningkat pada sekitar dua pertiga pasien. Kadar protein C-reaktif meningkat pada sebagian besar individu, tetapi cenderung menurun seiring dengan penurunan kadar troponin selama perawatan di rumah sakit.[19]
Apabila tersedia, dilakukan pemeriksaan untuk kemungkinan etiologi miokarditis dan perikarditis lain, terutama infeksi COVID-19 akut atau sebelumnya, dan virus lain seperti enterovirus.[19]
Pemeriksaan Pencitraan
MRI jantung menunjukkan temuan sugestif miokarditis, seperti peningkatan gadolinium dan edema miokard. Pada studi Das et al., 69% pasien menunjukkan adanya late gadolinium enhancement (LEG), dan 35% pasien tampak gambaran edema miokardium pada MRI T2 mapping.[19,29]
Tata Laksana Miokarditis dan Perikarditis Pasca Vaksinasi COVID-19 mRNA
Penting untuk melakukan tinjauan medis yang tepat apabila ada kecurigaan miokarditis atau perikarditis pada remaja atau dewasa muda setelah vaksinasi COVID-19 mRNA. Dalam evaluasi dan penanganannya, perlu melibatkan dokter spesialis jantung.[14]
Perawatan di Rumah Sakit
Evaluasi dan manajemen bervariasi, tergantung pada usia pasien, presentasi klinis, penyebab dan komorbid lain, stabilitas hemodinamik, dan perjalanan klinis. Data CDC menunjukkan bahwa 96% pasien dari 323 kasus terkonfirmasi miokarditis/perikarditis pasca vaksinasi dirawat di rumah sakit.
Pasien dengan miokarditis harus dirawat di rumah sakit untuk pemantauan jantung, termasuk pemeriksaan EKG berkelanjutan dan pemantauan kadar troponin secara serial. Namun, sebagian besar pasien dipulangkan dengan resolusi gejala.[14]
Medikamentosa
Data saat ini menunjukkan bahwa sebagian besar kasus miokarditis atau perikarditis pasca vaksinasi COVID-19 mRNA memiliki gejala ringan dan dapat pulih dengan baik. Informasi tentang gejala sisa jangka panjang belum tersedia. Terapi suportif dapat diberikan obat antiinflamasi nonsteroid (aspirin, ibuprofen, dan indometasin), metilprednisolon, dan kolkisin. Beberapa pasien ada yang diberikan imunoglobulin intravena.[19]
Jarang terjadi kasus berat yang membutuhkan manajemen khusus untuk aritmia dan gagal jantung kongestif, seperti ACE-inhibitor dan beta blocker. Tindakan mekanis pendukung lain, seperti pemasangan ventricular assist device, mungkin dibutuhkan untuk pasien yang mengalami gangguan ventrikel jantung.[19]
Penundaan Vaksin Dosis Kedua
Saat ini National Advisory Committee on Immunization (NACI) merekomendasikan agar individu yang mengalami miokarditis dan/atau perikarditis setelah dosis pertama vaksin COVID-19 mRNA menunda dosis kedua, hingga lebih banyak data penelitian yang tersedia.[13,31]
Larangan Aktivitas Berat
Meskipun perjalanan klinis dari miokarditis dan perikarditis menunjukan gejala ringan, pembatasan aktivitas fisik dan olahraga berat tetap diperlukan, termasuk berenang, lari, senam aerobik, dan bersepeda.[2,32]
Pembatasan ini dilakukan hingga adanya resolusi tanda dan gejala, hemodinamik, ritme jantung, hasil laboratorium, EKG, maupun pencitraan jantung. Pasien mioperikarditis yang akan melakukan olahraga harus berkonsultasi terlebih dahulu dengan dokter spesialis jantung yang merawat.[2,32]
Laporan kasus miokarditis dan perikarditis dalam minggu pertama pasca vaksinasi COVID-19 ini membuat kementerian kesehatan Singapura menganjurkan individu untuk menghindari aktivitas fisik atau olahraga berat selama 2 minggu pasca vaksinasi.[32]
Kesimpulan
Berbagai studi telah melaporkan efek samping miokarditis dan/atau perikarditis pasca vaksinasi COVID-19 mRNA, termasuk vaksin Pfizer dan Moderna. Walaupun mekanisme terjadinya belum diketahui secara pasti, tetapi kondisi ini juga telah dilaporkan sebagai efek samping yang jarang setelah penyuntikan vaksin influenza, vaksin hepatitis B, dan vaksin varicella.
Miokarditis pasca vaksin COVID-19 mRNA paling sering dilaporkan pada hari ke-3 setelah penyuntikan, sedangkan perikarditis pada hari ke-20. Sebagian besar terjadi setelah pemberian dosis kedua, dan pada laki-laki usia remaja atau dewasa muda (16 tahun ke atas). Pada sebagian besar kasus, pasien merespon baik terhadap tata laksana dan mampu mengalami perbaikan gejala yang cepat dengan istirahat.
Penelitian lebih lanjut diperlukan untuk mengetahui mekanisme patofisiologis, sehingga dapat mengidentifikasi atau mencegah kejadian di masa depan. Selain itu, dibutuhkan juga adanya informasi mengenai efek jangka panjang pada penderita miokarditis atau perikarditis pasca vaksinasi COVID-19.
Dengan pertimbangan manfaat, risiko, morbiditas, dan mortalitas infeksi COVID-19, bahkan pada individu remaja maupun dewasa muda, maka keputusan untuk pemberian vaksin COVID-19 masih tetap sangat bermanfaat. Jarak penyuntikan dosis kedua dari dosis pertama vaksin mRNA pada orang muda juga masih dipelajari, sebagai upaya untuk mengurangi risiko miokarditis dan perikarditis.